 You can use the questions to evaluate the presentations.ġ. When students bring their models to class, each group can come together to learn from each member’s attempt and put forth the best model or models for their presentation.Īs students develop their presentations and build their models, have groups answer the following questions. 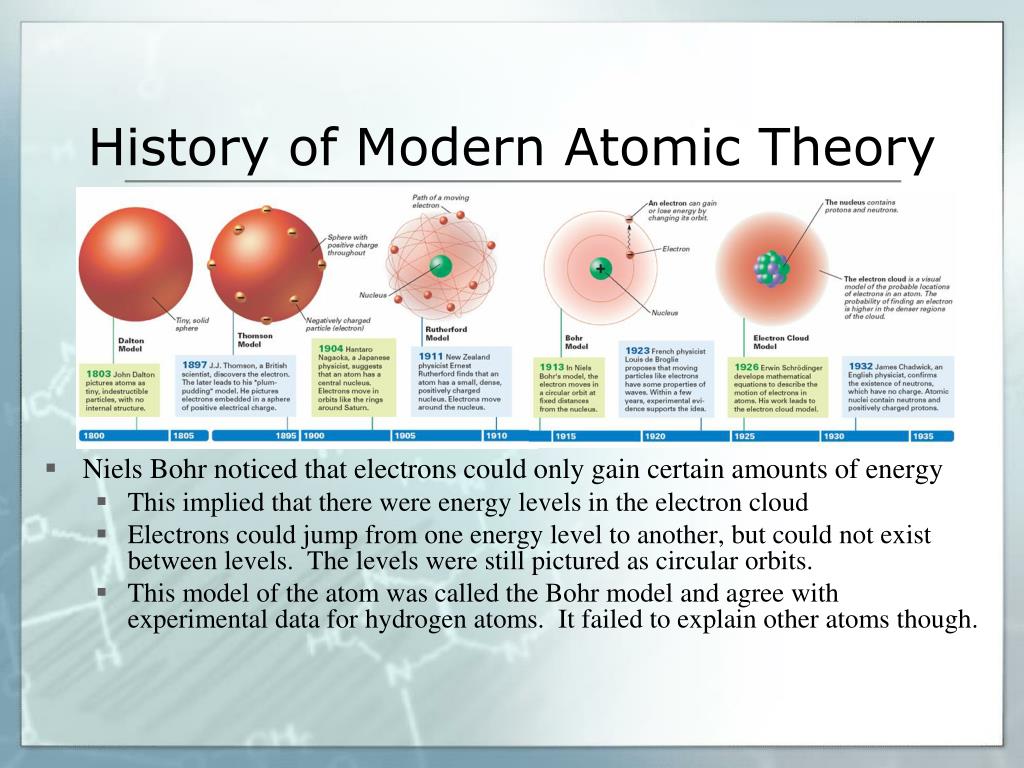
Alternatively, you could assign model building to students individually. Creativity is encouraged, but students should still aim for accuracy. Groups should build physical representations of their assigned models to incorporate in their presentations. Each presentation should include a description of the model’s appearance, information about the scientists that developed the model and what was known about atoms at the time the model was developed. The groups will work in class and if needed, at home, to research and prepare 5- to 7-minute presentations about their models, to be delivered at the next class. Assign one of the classic models - Dalton, Thomson, Rutherford, Bohr and Schrödinger - to each group. Knowing the structure of atoms also aids in the design of medicines and materials such as those used in environmental monitoring equipment and solar panels.Īfter reviewing the homework answers, divide the students into groups. For instance, chemists can predict melting and freezing points for chemicals they create when they know the structures of their starting molecules. Students may note that knowing the structure of atoms can help scientists determine how chemicals will react with each other. What are some practical implications of knowing the structure of the atom?Īnswers will vary. Why has the model of the atom changed over time?Īs scientists do experiments and make discoveries, they change their models to reflect new knowledge.ģ. Atomic structure helps scientists understand and predict the properties of matter.Ģ. Knowing how many protons, neutrons and electrons a particular atom has will tell you what chemical element it is and how it will behave with atoms of different elements. The basic components of atoms are protons, neutrons and electrons. What is an atom and why do we care about its structure?Ītoms make up matter they are the building blocks of chemistry. A version of the story, “Cracking the atom,” appears in the Apissue of Science News.ġ. “ Dig into atomic models” student worksheetīefore the first class for this activity, have students read the online Science News article “ How matter’s hidden complexity unleashed the power of nuclear physics” and answer the first set of questions on the student worksheet for homework. cotton balls, pipe cleaners, packing materials, yarn, tennis balls, baseballs, baggies and other items that can be found at home or in school) Various building materials (examples could include beans, candies. 
Want to make it a virtual lesson? This activity can be delivered remotely using meeting software that allows you to convene the whole class and also to divide students into groups. To develop their ideas, the class should draw on current scientific knowledge and use resources such as CERN: The Standard Model. In the final session for this activity, teachers will discuss the standard model of particle physics and have the students brainstorm ways they might create a 2-D or 3-D interpretation of it. During the second class, the groups can finalize their physical models and make their presentations to the rest of the class. For homework, students can work with their groups or individually to create a 2-D or 3-D physical model of their group’s classic model. To prepare for the first class, students will read the online Science News article “ How matter’s hidden complexity unleashed the power of nuclear physics,” and answer three questions for homework.ĭuring the first class, students will work in groups to research a classic atomic model and begin preparing a presentation about it. Procedural overview: This activity is designed for three class periods and includes two homework assignments. The class also will learn about the standard model of particle physics and brainstorm how it could be shown in two or three dimensions. 
Purpose: Students will work in groups to research, create and present different models of the atom that show the evolution of physicists’ understanding of atomic structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
